News 9/9/20
Top News

A federal court dismisses a class action lawsuit that was brought by a patient of University of Chicago Medical Center who objected that the hospital shares the de-identified records of adult patients with Google for machine learning research.
The plaintiff says the hospital shares patient dates of service and free-text medical notes, both of which could make it easier for Google to re-identify their data. He adds that the company could link his information to the many databases it controls, including geotracking information, which could compromise his anonymity.
The court found that while the hospital probably breached the express contract that is represented by its Notice of Privacy Practices by “selling” de-identified data in return for free software licenses, courts have not ruled that such data has market value. It also said that the plaintiff did not prove that the value of his records was diminished by having them shared with Google.
The court noted a legal precedent in which a HIPAA violation can’t be pursued as a breach-of-contract lawsuit.
The patient sought damages, an argument that the court found was not supported by proof of any losses. The ruling also observed that the plaintiff signed the hospital’s authorization form, which specifies that patients don’t get paid even if their information is used to create commercial value.
Reader Comments
From HITCurious: “Re: HIMSS20. The exhibitor settlement contract alludes to class action lawsuit. Anyone have details?” HIMSS provided a timely response to my inquiry, which I appreciate:
HIMSS confirms that in early June, one of its exhibitors filed a lawsuit in federal district court in Illinois, seeking a refund of fees for the 2020 Global Conference. This exhibitor also seeks to represent a class of other exhibitors that it claims are similarly situated. This lawsuit is captioned HatchMed Corp. v. Healthcare Information and Management Systems Society, Inc., Case No. 1:20-cv-03377 (N.D. Ill.). HatchMed alleges that HIMSS had an obligation to refund fees paid by exhibitors for the 2020 Conference, after the Conference was cancelled due to COVID-19. No class has been certified, and HIMSS disputes the basis for HatchMed’s claims.
HIMSS is grateful for the support of exhibitors and attendees through these unprecedented times, and gratified by the overwhelming support of exhibitors for the upcoming HIMSS21 conference, scheduled for August 2021.
[HatchMed, which sells nurse call cables and device mounts and reports 2-10 employees, booked a 10×20 booth for $11,075].
HIStalk Announcements and Requests
Reminder: if you have struck out on your own in health IT marketing/PR at some point in your career, share your experience with those who are considering such a move.
Webinars
September 9 (Wednesday) 1 ET: “APIs for Data Liquidity in Pandemic Times.” Sponsor: Chilmark Research. Presenters: Brian Murphy, research director, Chilmark Research; Gautam “G” Shah, VP of platform and marketplace, Change Healthcare; Drew Ivan, chief product and strategy officer, Lyniate; and Dave Levine, MD, co-founder and chief medical officer, Datica. This webinar will present the findings from a recently published research report on the state of the healthcare API market. The presenters will describe their work in deploying APIs to enable new functionality to address COVID-19. They will cover the use cases that have been most reliable for enabling effective data liquidity, how developers are using APIs to respond to the pandemic, and how different parts of the healthcare system are making APIs more widely available.
September 17 (Thursday) 1 ET. “ICD-10-CM 2021 Updates and Regulatory Readiness.“ Sponsor: Intelligent Medical Objects. Presenters: June Bronnert, MSHI, RHIA, VP of global clinical services, IMO; Theresa Rihanek, MHA, RHIA, mapping manager, IMO; Julie Glasgow, MD, clinical terminologist, IMO. IMO’s top coding professionals and thought leaders will review additions, deletions, and other revisions to the 2020 ICD-10-CM code set that will be critical in coding accurately for proper reimbursement.
Previous webinars are on our YouTube channel. Contact Lorre to present your own.
Acquisitions, Funding, Business, and Stock
ConnectiveRx invests in HelpAround, a patient engagement and medication adherence startup that is focused on app-based messaging for patients undergoing specialty treatments.
People

Access EForms promotes Rob True to VP of professional services.


Collaborative Imaging names Arun Douglas (Zinc) chief technology and security officer and David Silva (Baylor Scott & White Health) chief compliance officer.



Health Catalyst co-founder Steve Barlow returns as SVP after a three-year missionary stint. Bryan Hinton will be promoted to CTO in replacing Dale Sanders, who will move to senior advisor.

Australian Digital Health Agency hires Amanda Cattermole, MS, MBA (Services Australia) as CEO. She replaces Tim Kelsey, who took an SVP job with HIMSS in January 2020.
Announcements and Implementations
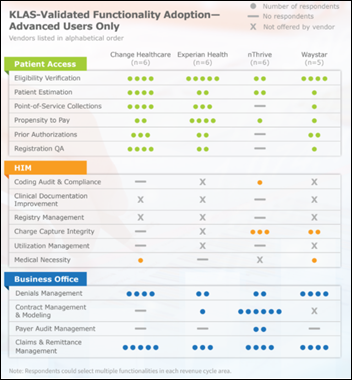
A new KLAS report finds that health system use of a single vendor for all revenue cycle needs remains a work in progress, with clients of Change Healthcare and Experian Health reporting the highest usage of available components as the slow transition from best-of-breed solutions continues. Waystar clients say they want to implement available patient access functionality, those of NThrive say its technology can handle complexity, and both companies have multiple customers using HIM functionality that has limited industry-wide enterprise use.
COVID-19
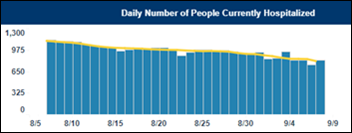
North Carolina becomes the latest of several states for which published COVID-19 numbers have been skewed due to data reporting problems. The state’s HHS says that unspecified technical issues have caused hospitalization data to be underreported since Friday.

Stat assesses the progress that has been made by Operation Warp Speed in its first five months:
- It spent $10 billion to support vaccine manufacturer research and production.
- It spent $450 million to support the monoclonal antibody treatment manufacturing capacity of Regeneron (and soon to Eli Lilly, which is working on a similar product). This is its only investment in COVID-19 treatment.
- It has done little with diagnostics.
- It has issued $500 million worth of no-bid contracts with companies that manufacture syringes and glass vials.
- Stat gives OWS low marks for transparency, such as explaining why it chose particular vaccine projects to fund.
Economics researchers estimate that the 500,000 attendees in early August’s Sturgis, SD motorcycle rally — most of whom did not wear masks or use social distancing — created 250,000 new coronavirus cases that will incur $12 billion in public health costs.
A pre-print of a randomized study of 464 hospital inpatients by India-based researchers finds that convalescent plasma treatment was not associated with reduced mortality or a halt in disease progression. The authors note that while CP therapy is authorized for off-label use in India and is safe, it requires a lot of resources to collect and store plasma and its sale on the black market has driven prices up.
The Washington Post says that businesses that perform “deep cleaning,” spray disinfectant freely, and make employees wear gloves is just “sanitation theater” that may distract from the more important need for people to socially distance, wear masks, and avoid enclosed spaces. it may also provide a false sense of security in making it appear that the virus can be controlled.

A pathologist employee of Texas-based freestanding ED operator Physicians Premier ER quits after the company bills his insurer $11,000 for a COVID-19 antibody test. The doctor, who ironically oversees the company’s COVID-19 antibody testing program, was a temporary employee between jobs whose short-term “junk insurance” plan was issued by United Healthcare-owned Golden Rule. ED staff told him that he wouldn’t have to pay anything. Golden Rule paid $2,100 without questioning the total, but then the facility sent him a bill for $8,900, which included services he says he didn’t receive in his 30-minute visit. The company’s advertising says the test costs $75 and Medicare pays $42 for it. The doctor reported his concerns to Golden Rule, whose investigator told him he wasn’t surprised and then didn’t follow up.
Other

A local news outlet looks at the impact observational studies that are published through Epic’s recently launched Health Research Network have had on COVID-19 treatments:
- Data queried by researchers at Lee Health (FL) showed that COVID-19 patients were three times as likely as others to develop blood clots, which led doctors to routinely give blood thinners to high-risk patients.
- Mount Sinai Health (NY) researchers found that patients in their 60s and 70s with dementia were more likely to be hospitalized or die from COVID-19 than patients the same age without dementia.
- Epic’s own analysis of COVID-19 patients with high blood pressure found those on RAAS inhibitors did no worse than those on other drugs, and did better than those not taking any blood pressure medication.
- The company’s months-long analysis of cancer screenings has found that, though they have rebounded from significant drops in the spring, they’re still one-third lower than normal.
Sponsor Updates
- Arcadia publishes a new case study, “CareMount ACO Uses Arcadia Analytics to Build a Narrow SNF Network and Reduce ALOS by 4 Days.”
- Collective Medical enables SNFs to rapidly generate reports on COVID-positive patients and report to CMS.
- ESolutions publishes a new white paper, “How the RAP Phase Out will Affect HHA Billing Requirements.”
- Everbridge risk intelligence provides major corporations, healthcare organizations, and government entities with situational awareness to mitigate threats from COVID-19.
- Health Catalyst completes its seventh annual and first ever virtual Healthcare Analytics Summit, with record registration of over 3,500 attendees.
Blog Posts
- How to Create Health EHR Interfaces (314e)
- Is telehealth right for your practice: Key considerations for physicians interested in virtual care (Ability Network)
- The Access Points Podcast: Tools, Habits, & Ideas for Peak Performance (Access)
- Creating the Visually Sustainable Healthcare System (Agfa Healthcare)
- Demo: Epidemiology and Epidemiology Rate Dashboards (Bluetree)
- Saratoga Hospital Uses Vocera Solution to Prepare for COVID-19 and Improve Staff Safety (Vocera)
- The Right Medical Device Integration Strategy can Increase the Efficiency of Medical Device Security (Capsule)
- Tips and Tricks for New VSAC Authors (Clinical Architecture)
- Managing Chronic Disease: 3 Steps Payers are Taking (Collective Medical)
- What Patients Want Out of Healthcare is Changing (CoverMyMeds)
- How COVID-19 Contact Tracing Works (Dimensional Insight)
- Care Traffic Control: Monitoring Patient Progress at Home (Dina)
- Back to the Basics During the COVID-19 Era (EClinicalWorks)
- We Owe it to Patients to Eliminate Faxing: Here’s How to Get Started (Ensocare)
Contacts
Mr. H, Lorre, Jenn, Dr. Jayne.
Get HIStalk updates.
Send news or rumors.
Contact us.




RE the AI GLP1 company, Washington Post has an article today by someone who used one of those compounded products,…