The UHC hiding the breach notice page is one thing but it looks like Nebraska is suing Change Healthcare for…
Monday Morning Update 3/24/14
Top News
Three Detroit hospital systems – Beaumont, Oakwood, and Botsford – announce plans to merge into an eight-hospital, $3.8 billion system, citing shared electronic medical records as one of their four goals.
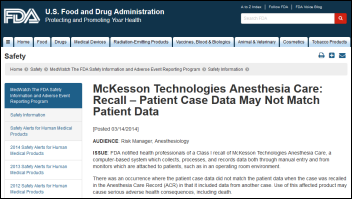
From Tom: “Re: McKesson’s FDA Class 1 recall. The description of their product Anesthesia Care could generically be applied to almost any EMR/EHR/CIS vendor’s AIMS product and yet the FDA’s decision-making clearly does not apply to vendors equally. Also I wonder how the regulation of CDS would affect a hospital who develops their own CDS?” FDA’s highest-level recall of McKesson Anesthesia Care may be sending a message that the agency considers even software-only clinical decision support to be high risk. McKesson defines its product as an anesthesia information management system, which it also calls an “anesthesia EMR.” McKesson sought and received FDA premarket clearance apparently because the system collects data from physiologic monitors. McKesson did a voluntary recall of its product in March 2013 after a customer reported that the software pulled up the wrong patient’s information, with two other customers reporting later that it had lost medical history comments and misconnected to a physiologic monitor, affecting one patient in each instance. Some thoughts:
- McKesson Anesthesia Care is a software-only system that does not control medical devices. It collects and uses information from patient monitors. Other than that, it’s like any other high-acuity, unregulated EHR (surgery, ICU, ED, etc.)
- FDA would not have been involved if the patient monitor connection hadn’t pushed the product into its regulatory arena. FDA regulates software that makes independent patient decisions or connects to regulated devices, with the idea being that those systems are devices working on their own rather than simply providing guidance to users.
- Software vendors usually hide contractually behind the “professional judgment” test that says even if their software gives incorrect information or bad advice that harms patients, the clinical professional who uses the system makes the final decision and is solely responsible for the result.
- The danger to patients is the same as for any other clinical decision support or even EHR software. Mixing up information between patients could be disastrous any time software is presented information or recommending actions. However, high-acuity systems give users less time to make important decisions, so that probably should be a consideration in determining patient risk.
- McKesson planned to announced a Class II recall (meaning the problem wasn’t likely to cause patient harm) but FDA overrode that proposal and initiated a Class I recall indicating that patients could be harmed.
- McKesson notified users almost immediately when the first problem was reported in March 2013, but FDA’s recall didn’t go out until a year later.
- It’s not clear what users of the system should do as an alternative, or what action they may have taken since the original McKesson notification last year.
- Vendors of systems that perform equally critical functions that aren’t connected to medical devices can take whatever action they want if they are faced with the same problem since their software isn’t regulated by FDA. Other than to avoid legal exposure, they could arguably not inform customers at all.
- McKesson is a member of the HIMSS Electronic Health Record Association, a trade group that requires them to sign the EHR Developer Code of Conduct asserting, “We will notify our customers should we identify or become aware of a software issue that could materially affect patient safety, and offer solutions.” The other inpatient EHR vendor members are Allscripts, Cerner, Epic, GE, NextGen, and Siemens.
- McKesson backed legislation introduced last month (along with athenahealth, IBM, and trade groups) that would reduce “unnecessary regulatory burdens” by limiting FDA’s oversight of “low-risk health IT, including mobile wellness apps, scheduling software, and electronic health records.”
- FDA is running late in producing a report that it says will explain its position on regulation of clinical decision support systems.
From LochnessMonster: “Re: McKesson. Reduction in force 3/20/14, roughly 300 under Pat Blake organization (uncertain number).” Unverified, but reported by multiple readers, one of them saying that the targeted areas were Horizon and Paragon.
From Bootay: “Re: vendor-convened panels. You should participate or report the results.” I don’t think so. I’ve seen many times where properly objective people turned into fawning, attention-starved glad-handers just because some company tries to buy their love by inviting them to be a speaker or advisor. It makes my skin crawl to see the obvious mutual sucking up as mutually expectant backs wait to be scratched.
HIStalk Announcements and Requests
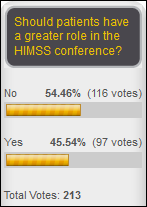
A slight majority of respondents don’t think patients should have a greater role in the HIMSS conference. New poll to your right: who’s most responsible for the problems with health insurance exchanges?
Welcome to new HIStalk Platinum Sponsor ScImage (pronounced sye-image). The Los Altos, CA-based imaging and informatics company offers solutions that include enterprise imaging, radiology, cardiology, Echo PACS, ECG, cloud PACS, EMR content management, vendor-neutral archive, and a Web-based DICOM exchange. Case studies include Missouri Baptist Medical Center’s cardiology PACS, Blessing Hospital’s enterprise PACS, and US Air Force’s cardiology consultation program. The privately held, employee-owned, debt-free company says it has never sunsetted a product or required a forklift upgrade. According to a physician at Cedars Sinai Heart Institute, the company’s products are the “ultimate value proposition” to its cardiology practice. Thanks to ScImage for supporting HIStalk.
Here’s ScImage PACS consolidation overview I found on YouTube.
Teach for America teacher Ms. A sent pictures of her students using the Chromebook that we as HIStalk readers provided to her first grade classroom in Maryland via DonorsChoose. They’re using it to access online reading and math programs.
Upcoming Webinars
April 2 (Wednesday) 1:00 ET. A Landmark 12-Point Review of Population Health Management Companies. Sponsored by Health Catalyst. Presenter: Dale Sanders, SVP, Health Catalyst. Learn the 12 criteria that a health system should use to evaluate population health vendors and to plot its internal strategy, then see the results of grading seven top PHM vendors against these criteria. No single vendor can meet all PHM needs. The most important of the 12 criteria over the next three years will be precise patient registries, patient-provider attribution, and precise numerators in patient registries.
Acquisitions, Funding, Business, and Stock

WelVU, which offers a personalized patient education application, raises $1.25 million in an initial seed round.
People

Effingham Health System (GA) promotes Mary Pizzino to CIO.

CHIME promotes Keith Fraidenburg to EVP/chief strategy officer.
Announcements and Implementations
Physicians at Jupiter Medical Center (FL) are piloting the use of email alerts and status updates when their ACO patients are seen in the ED or urgent care center. The press release is poorly written and the product has a confusing name: MicroBloggingMD. I saw their booth at HIMSS and thought it was yet another doctor writing a blog.
Connecticut officials say Massachusetts owes the state $10 million of the $45 million in federal money it received to build its struggling Massachusetts Health Connector. The original grant called for Massachusetts to share its technology plans with other New England states, but those other states realized they could get their own federal money for building exchanges and went their own way, with Connecticut receiving $140 million, Rhode Island $113 million, Vermont $168 million, and Massachusetts a total of $179 million. Massachusetts says the money wasn’t intended for the other states – they were added on to the grant application at the last minute after pressure from the White House and Governor Deval Patrick to make Massachusetts a model for the rest of the country. Access Health CT’s CEO says that unlike the dysfunctional, CGI-built Massachusetts exchange, their Deloitte-created one works fine, adding, “Some states were trying to build a Maserati. We built a Ford Focus. It might not be as glamorous, but it runs. It can get you to the store.”
Google is a bit touchy over Google Glass, having previously urged its users to avoid the “Glasshole” label by not being “creepy or rude.” Now it shares “The Top 10 Google Glass Myths,” the one above being notable considering that people (some of them Glassholes, no doubt) are already using it in patient care. Google published the statement on Google Plus, which means almost nobody other than its own employees will see it.
Other

Duke University Health System (NC) will pay $1 million to settle charges that it overbilled the government by unbundling claims and billing for PA services in heart surgery. Duke says its mistake wasn’t intentional, but instead “resulted from an undetected software problem and through possible misapplication of certain technical billing requirements.” A former Duke employee had filed the whistleblower lawsuit.

In England, the local newspaper reviews the 2012 Meditech go-live at Rotherham NHS Foundation Trust that caused delays in cancer treatment, lost appointments, and cost the hospital $2 million in revenue. It mentions the project review, which found that delivery targets weren’t specific, penalties clauses were vague, and the 18-month timetable was unrealistic given that the system had never been implemented in the UK. Taxpayers got stuck with $17 million in cost overruns on top of the budgeted cost of $49 million.
A two-doctor cardiology practice in Texas will pay $3.9 million to settle Medicare fraud charges for conducting unneeded procedures. Authorities requested data from 100 nuclear tests that had been performed, but the doctors provided only 37, saying their computer had crashed and the other results were lost. The investigators found that 19 of the 37 tests had been interpreted incorrectly and 75 percent of them were performed wrong. The same foreign-born doctors were part of a group that settled for $27 million in a 2009 Medicare fraud case.

Stanford Hospital & Clinics and its former collections agency are expected to pay $4.1 million to settle charges that the information of 20,000 ED patients was posted online for nearly a year. Stanford says it encrypted the information sent to the agency, but that company forwarded it to get help creating a graph and the worksheet ended up on a student homework site.
A Motley Fool review of mobile health in China, which a Brookings Institution report says will be worth $2 billion per year by 2017, says the three publicly traded companies that will benefit most are IBM, Microsoft, and Lenovo. It says the market won’t behave as it does here because Chinese medicine has different workflows, the language is hard, cloud-based security is a tough sell, and Apple’s mobile devices are much less popular than Android ones. It misses some facts: (a) most mHealth companies aren’t publicly traded; (b) those three companies are so large that whatever happens with mHealth in China isn’t going to move the share price; (c) it touts Microsoft as having implemented “a single, cloud-based system” that turns out to be the nearly forgotten HealthVault; (d) it predicts Lenovo’s success because it makes hybrid devices (laptop/tablet) that run Windows 8 and because it bought Motorola and found itself owning 11.8 percent of the smartphone market in China, although the article fails to mention Lenovo’s huge benefit: it’s a Chinese company.
Sponsor Updates
- Health Data Specialists will exhibit at the Cerner Southeast Regional Users Group March 30 – April 4 at the Sheraton Sand Key in Clearwater Beach, FL.
Exhibitor Costs at the HIMSS Conference

Readers had asked for details on what it costs a company to exhibit at the HIMSS conference. I greatly appreciate the vendor executive (let’s call him “Larry,” just to keep things anonymous) who provided complete information from last month.
Booth construction: $132,000
Booth space (20×40): $26,000
Booth power and connectivity: $20,000
Breakfast briefing: $11,000
Hospitality suite: $15,000
Printing: $6,000
Giveaways: $4,000
Booth graphics: $2,500
Buying the attendee list: $1,800
Including some other smaller costs, the company’s total expense was $222,000. That doesn’t include employee salaries or travel costs.
Larry says he’s happy with the outcome. The company had 400 people visit the booth for meetings or to see a demo. About half of those had been scheduled in advance, which is an efficient way to meet with prospects, and the other 200 were walk-ups who might become prospects. He also sees value in the employee bonding experience and being able to learn from attendees.
It’s the same as for attendees, in other words: HIMSS benefits from putting interesting people in the same place at the same time. The attendees derive their value from each other.
Contacts
Mr. H, Inga, Dr. Jayne, Dr. Gregg, Lt. Dan, Dr. Travis, Lorre.
More news: HIStalk Practice, HIStalk Connect.







RE: Cost to exhibit at HIMSS
Yikes! Booth power and connectivity: $20,000
I guess I won’t complain about my Comcast bill anymore! But that seems excessive to me for electricity and internet for a few days. Seems like gouging to me.
Overall, about a quarter of a million dollars to be a vendor at HIMSS. hmmmm.
How is having Dale Sanders give a seminar on the twelve key things to look for in a pop health system anything other than a commercial for Health Catelyst ???
He’s notoriously critical about everyone other than himself.
Wonder how HC will stack up to his model.
“McKesson is a member of the HIMSS Electronic Health Record Association, a trade group that requires them to sign the EHR Developer Code of Conduct asserting, “We will notify our customers should we identify or become aware of a software issue that could materially affect patient safety, and offer solutions.” The other inpatient EHR vendor members are Allscripts, Cerner, Epic, GE, NextGen, and Siemens.”
Are you certain that all of those vendors have committed to this and all of the provisions of the EHR Developer Code of Conduct? Is there a list of which vendors have committed to which (all) provisions?
Here’s what the EHRA site says (which is much less ironclad about commitment): http://www.himssehra.org/ASP/codeofconduct.asp
Recognizing that every company has different business models and practices, companies that adopt the Code will be responsible for determining their specific application of the Code consistent with the spirit of the Code’s principles. Adoption of the Code means that the company is committed to all of its principles and has completed or is underway in the implementation of the practices needed to apply these principles within the company
Ron
Regarding the Detroit hospitals,Beaumont, Oakwood, and Botsford and one of their goals, the EHR, that is not a reason to merge! Any organization that uses technology as a reason is doomed for failure. With all that has been written about the initial cost and sustainability of EHR, really that is a goal? I would like to see what their other goals would be, maybe centered around the patient and costs? Sad way to publicize the merger.
Hey Vendor briefing…
The webinars are PAID advertisements…what you expect?
and over in another blog Saunders is listed a CIO of a Cayman Island facility. Unfortunately it looks like all the blogs are doing this. Get vendors to pay and trump up a webinar. I think Mr. H should require webinar presenters to sign off on a conflict of interest disclosure and make it avail to webinar registrants. At least we’d have a better idea of where they are coming from.
Seems you can’t tell the players without a scorecard!
Waiting impatiently to hear if McKesson was really dumb enough to lay of staff in the Paragon business unit. The product suite is woefully behind Epic and Cerner in functionality, although THEY don’t think so. If true, this may hasten our exit from the platform.
@histalk : “McKesson Anesthesia Care is a software-only system that does not control medical devices. It collects and uses information from patient monitors. Other than that, it’s like any other high-acuity, unregulated EHR (surgery, ICU, ED, etc.)”
Hang on! Not exactly, if you read the F D and C Act’s definition of a medical device. If you decry the FDA stepping up to the plate to root out the widespread EHR dysfunctions, who, may I ask, do you think should perform that function?
To clarify re: the FDA’s scope, they actually have legal authority to regulate all EHRs and other similar software applications, however they have previously exercised what they call their “regulatory discretion” and opt to not actively regulate these applications. They do, however, reserve the right to reverse this thinking at any time.
This is why some members of Congress have recently proposed regulation to explicitly remove FDA’s regulatory authority over software. People who want to get into this space as a business do not like relying on the regulatory uncertainty this creates.
DrM – the oft-repeated meme that “regulatory uncertainty” of health IT by the FDA is stifling entrance into this market seems unsupported by the entrance of thousands of new EHR vendors, tens of thousands of new mobile medical app vendors and billions of dollars into this market.
Or that the bills removing FDA authority over software (!) will result in the absolute certainty that those on the sidelines allegedly require to invest.
A better, more productive discussion is one that involves rapid and substantive steps by vendors and their customers to ensure that health IT is built, implemented and used in ways that don’t endanger patient safety.
Completely agree with you Ron. I think it’s political theater myself.
RE: McKesson –
I install Anesthesia systems and educate anesthesia providers on the use and care of their EHR and Biomedical Device data. What is important to note as an anesthesia provider is that you are not ‘Monitoring’ your patient’s status off of your EMR. That is what your physiologic monitors are for. If a patient has a bad outcome and you say ‘well the data in my EMR told me otherwise’ then shame on you. Look at your ;monitors, anesthesia machine displays and your patient. It’s a synchronous event unlike other users of the EMR. You’re there with your patient throughout the procedure.
Your EMR is a data repository for what ‘has happened’ with the patient, should be validated regularly by you, the clinician to agree that it is correct and can be used for alerts and clinical decision support but does not replace your clinical judgment.
As for wrong patient/wrong record – that is possible with other systems and again, requires the appropriate safety checks by the IT/Biomed teams and the CLINICIAN!